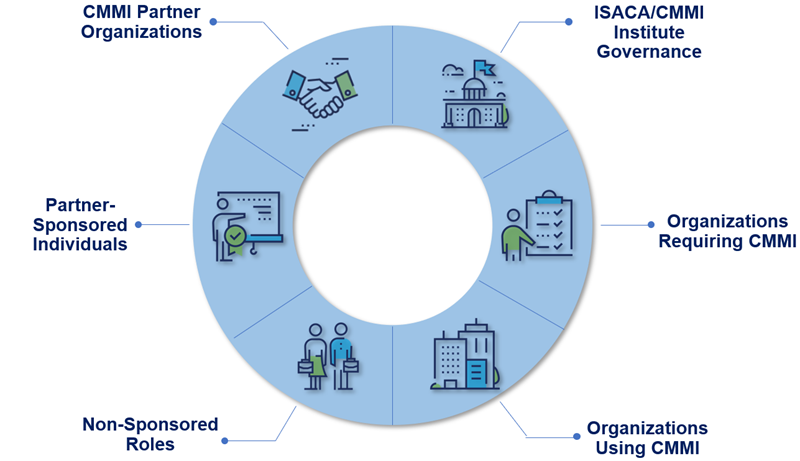
Here are other roles in the CMMI ecosystem that are not necessarily affiliated with a CMMI Partner:
Registered Interpreters support CMMI appraisal or course activities with translation services.
CMMI Associates are committed to excellence in capability improvement and can connect the CMMI Model and appraisal process with business value and use the model as a tool for improving performance. They can participate in a CMMI Appraisal as an Appraisal Team Member and are certified by ISACA performance improvement initiatives, provide organizational roadmaps and lead ongoing organizational change efforts.
CMMI Professionals are experienced process, performance and quality individuals who coach organizations on performance improvement initiatives, provide organizational roadmaps and lead ongoing organizational change efforts. A CMMI Professional has achieved CMMI Associate status already and is certified by ISACA.
Consultants guide organizations to create a performance improvement program to help it meet strategic business objectives. ISACA does not train, certify or otherwise vet CMMI consultants at this time.